|
|
GENERAL EQUIPMENT INC.
8724 Sunset Drive #191● Miami FL 33173 · USA
|
|
|
GENERAL EQUIPMENT INC.
8724 Sunset Drive #191● Miami FL 33173 · USA
|
IV FACILITY
|
TYPE: |
Medical Supply |
|
DESCRIPTION: |
IV Facilty |
|
PRICE: |
Upon Request |
|
PACKING: |
Upon Request |
iv facility
From New Manufacture

Construction a facility in for the production of IV solutions to be used for patients
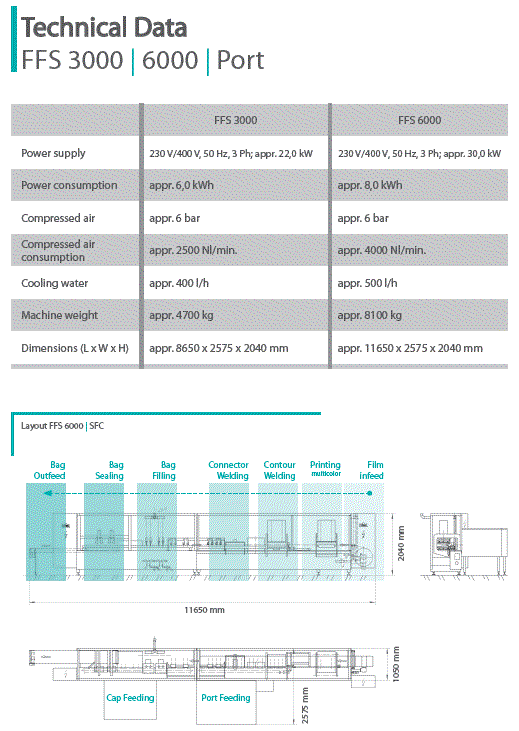
The facility will use the Plumat Form Seal model FFS 3000 system that can produce bags ranging from 50 to 2,000 mL with the standard bag being 500 mL
The proposed installation will produce 2,500 units per hour, (50, 100, 250 and 500 mL sizes with the larger sizes being less)
If the plant runs 8 hours per day for 250 days the annual production is 5,000,000 units
If the plant runs for 16 hours per day for 250 days the annual production is 10,000,000 units
If the plant runs fro 23 hours per day for 250 days the annual production is 14,375,000
Infusion Fluid Production Facility
Manufacturer is pleased to submit this proposal for the design, manufacture, installation, training, technology transfer, documentation, validation and start-up for a self-contained Medical Solutions Production Center for producing a wide range of Intravenous Medical Solutions (IV Fluids)
Utilities and infrastructure facilities like water, fuel gas, electricity, drainage, common effluent treatment plant, communication system, roads, should be available at the site.
The needed building is projected to be 2,100 square meters in size, (approximately 22,000 square feet on one floor, and is not part of this proposal but can be supplied at an additional cost. See below on building specifications.
The scope of the bid includes complete design and detailed engineering for the production building and the production area, procurement and supply of the state of art materials & machinery, mechanical erection, pre commissioning and commissioning, obtaining necessary clearances, for all components, systems and process equipments, project management, formulas and total process validations, supply and storage of necessary spares for the specified period, guarantee run, training of operating staff, and handing over the facility at the end of the specified period after the necessary performance tests are carried out and the plant is approved by such agencies which are deemed fit for approval, of IV Solutions production plant to be established in . This proposal does not include real estate, building structure, electrical power, drainage, sewage disposal and general infrastructure. The owner is responsible for all permits, fees and taxes that may be required by the local, state and federal governments and are not included in this proposal.
Production of LVP Solution products meeting European (EU) Pharmacopoeia quality, Good Manufacturing Practice (“GMP”) and ISO 9001:2000 standards.
Barring civil disorders or acts of God, the Project will be complete in 12 – 16 months. An average project can be completed within 13 months; however, the delivery time for some major items may push the project completion out to a longer time requirement. We will notify the owner of any delays once that they have been confirmed in writing by the supplier.
The technology and production facilities equipment, machines and systems will be provided. While there are several manufacturers of the different types of equipment needed for this project, there are no manufacturers that will supply and assemble all the necessary systems into a working facility. Manufacturer will include the technology transfer, provision of complete production facility, technical support for project implementation, technical and management support for pre-production.
The primary product line of the plant will be the standard intravenous solution, which contains pure water for injection and low concentrations of carbohydrates and salt, typical solutions and costs are shown below.
|
The production facility will include standard molds to manufacture containers of 100, 250, 500 and 1,000 mL.
These Products will be manufactured to the latest international standards and shall conform to all USFDA, cGMP, and ISO 9001:2000 regulations as well as any special regulations from local.
All the Equipment, Installation and the premises will be according to GMP Prescriptions, and all local applicable regulatory requirements.
The bags are supplied with a single or double SCF port for connection to an administration set. The bag has an overwrap applied before going into the sterilization process.
Based on the preferred production schedule described in the tender, the facility will have an annual capacity of 3.9 to 18,000,000 units of IV solutions per year depending on sizes and running time. The normal production rate is 2,500 units per hour of the 50,100,250 and 500 mL containers and 1,700 per hour at 1,000 mL
The IV
production facility has been designed to allow for the local
production of almost any type of IV solutions at a quality level
that exceeds the International Standards for IV solutions.
Water Supply
The facility can use
the water from the local supply system of the city, if
available, or it can come from a well driven on the property.
Before any design work is done on the water treatment process,
the raw water is tested for over 130 compounds. These tests must
be conducted on a routine basis to assure that the quality of
the water coming into the facility remains constant and that the
pre-treatment of the water is designed correctly. If a change in
source water quality is detected during testing, adjustments to
the Pre-Treatment process should be made by the plant laboratory
staff.
Pre-Treatment
Pre-Treatment of the
source water is composed of a series of filters that can perform
a number of functions including the removal of particulate
matter (including large bacteria), softening of water, and
adjustments of pH. Removal of some minerals may also occur in
this process. Each Pre-Treatment process is specifically
tailored as required by the quality of the source water. The
Quality Assurance program requires the plant laboratory staff to
test the output of the Pre-Treatment process every day that
Water for Injection (WFI) is made.
Reverse Osmosis
Reverse osmosis (R/O), also known as hyper filtration, removes of particles as small as dissolved individual ions from a solution. R/O is used to purify water and remove ions and dissolved organic molecules. R/O uses a membrane that is semi-permeable, allowing the fluid that is being purified to pass through it, while rejecting the contaminants that remain. Most R/O technology uses a process known as cross flow to allow the membrane to continually clean itself. As some of the fluid passes through the membrane the rest continues downstream, sweeping the rejected species away from the membrane, in concentrated brine reject water. The process of reverse osmosis requires a driving force to push the fluid through the membrane to reverse natural osmotic flow, and the most common force is pressure from a pump. The higher the pressure, the larger the driving force. As the concentration of the fluid being rejected increases, the driving force required to continue concentrating the fluid increases. Please note that the “reject” water will be used for “domestic and sanitary water” within the facility for toilet flushing, washing and other functions
Pure Steam Generator
Helps to purify the WFI, as well as provides the steam needed to clean and sterilize the machinery and to maintain the temperature level of the solutions in the systems.
Water Piping System
This system of
stainless steel pipes with polished interior walls deliver the
WFI water to a consolidation point where the WFI and chemicals
are mixed before entry to the Form Fill Seal machines in the
class 100,000 clean room. All the valves and pumps that are
involved are electronically monitored by the quality control
system of the facility.
Batch/Holding Tanks
The IV production will have two 30,000 liter stainless steel batch tanks to contain the full production of the facility for the full 23 hour, or less, production cycle. As one tank is drained for production the other tank will have been cleaned and refilled getting it ready for the next days production.
Form/Fill/Seal Equipment
The
FFS equipment is located in a class 10,000 clean room but has a
secondary soft wall that separates the rear of the machine from
the main clean room. This is installed for several reasons,
including sound level control, isolation of the hydraulic lines
from the front of the clean room, (if one should rupture the
operator will only have to clean a small area and no products
would be contaminated), and finally better heat control in the
clean room. Coming into the FFS units, above the ceiling of the
clean room, are pipes carrying the WFI with its added chemicals
to the machine. As stated before, there are also steam lines
coming to the FFS for the clean in place steam and sterilization
processes.
Included in the
FFS units is various electrical, optical and mechanical testing
or inspection points where the product is inspected as it is
being produced. The system will alert the operator of any
detected problems so corrections can be made quickly.
Label Application
The IV production systems print the label information directly onto the containers as part of the production process. Each label consists of information for users or consumers and a bar coded portion for tracking of the product to meet international standards. Exact label content will depend upon government requirements.
Overwrap
A protective overwrap is placed over the entire IV bag, this is an option and may not have to be done on all production runs. This is an automatic process.
Autoclaves
The filled bags
are placed into the autoclave on specially designed rack and
cart system. The products are terminally sterilized at high
pressure and temperature, 2 atmospheres at 121 C, in the
autoclave for the time required by the required national
standards. This time can vary from 12 to 20 minutes once the
proper temperature is reached. The autoclave has and access door
inside of and outside of the clean room, and acts as the product
pass point out of the clean room. All the temperature and time
data on each autoclave cycle is automatically recorded in the
plant’s computerized Quality Assurance records. By design, both
doors on the autoclave cannot be opened at the same time, thus
always insuring that the clean room environment is always
maintained. After the autoclave process, the empty racks are
returned to the autoclave where they are flashed sterilized
before being reused in the clean room.
Packaging
The
bags of IV solution and bottled water are packaged in cardboard
boxes for shipping. The owner must determine the count for each
box, 12 units per box are suggested but 24 units per box maybe
required larger clients. Each box is bar coded with the type of
product in the box, date of manufacturer, batch number,
expiration date, and the number of containers. This allows for a
simple record keeping system, accuracy in shipping, and accuracy
in billing. When each box is filled with the correct number of
units the box is sealed, using a semi-automatic tape system. The
full boxes are placed on a pallet for movement into a quarantine
storage area. This storage area is a designated spot in the
production section of the warehouse where the production batches
are stored for a 14 day quarantine period. The product is moved
to the designated warehouse area using either a manual or
electric jack/lift unit.
Warehouse
The warehouse is
set up to fully capitalize on different technologies utilized in
the total production process. This record keeping system is used
for government audits, quality control on the final products and
customer data. When product is shipped, the order is received
from the sales department at the warehouse desk, where it is
printed out. The print out includes what product type, lot
number and location of the solutions plus the shipping label(s)
for the boxes and packing list. This action automatically
adjusts the inventory system prompting additional production
activity.
All materials
coming into the facility from outside sources is recorded via
the computer system, which matches the material received with
what was ordered, and designates where the material is to be
stored until needed. This material includes plastic resins,
component parts, boxes, paper goods, chemicals or other items.
The receipt of any item that is used in a product will trigger a
work request for the laboratory to perform quality assurance
testing on the material ensuring that it meets or exceed the
specifications on the purchase order and the facility quality
assurance documentations. The material is only moved to storage
when it has passed the QA testing. If the material fails to pass
the testing, it is returned to the vendor for replacement. As
part of the tracking system, each container of material will be
labeled with the work order number that was used to test the
material.
Computer Network
The computer
network will be a wireless system, based off a Windows server.
Terminals will be located in selected areas, such as the
laboratory, finance, administration, and shipping/receiving.
All systems will be password protected so data will not be
compromised. Printers will be also located where needed. The
computer display can be in the local language or in English.
Manufacturer will have a connection to the computer system so
the quality programs can be monitored as part of any ongoing
agreement between the owner and the manufacturer.
The Quality Assurance Laboratory
The Quality
Assurance Laboratory will be equipped with the necessary
analyzers, incubators, microscopes and other equipment required
to maintain the ISO 9000 standards plus all purity requirements
of the various governmental departments.
The QA programs that are carried out by
both the laboratory and the manufacturer will meet or exceed all
international standards for quality, and will be the cornerstone
of the marketing program for the solutions both in and outside
of the country.
Unlike the IV
production plants of other designs, the plant limits the amount
of product handling by workers and vastly reduces chances of
contamination getting into the manufacturing process.
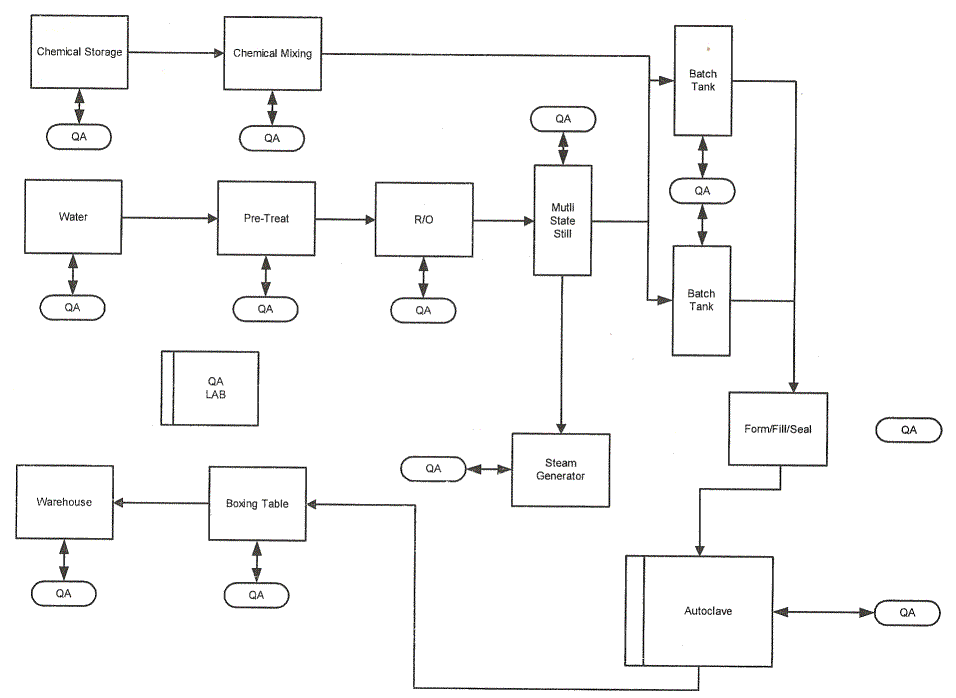
Chemical Storage
This secure space is used to store at least 5 days of chemicals for production. These chemical containers have all passed the QA inspections and are held in a secure area ready to be used in the production process. As the chemicals are removed from the area for mixing, new chemicals are brought into the area for future use only after passing the QA testing.
Bulk Storage
In this area, the plastic resins, a 30 day supply of chemicals, and a 15 day supply of boxes are kept. By limiting the space used for this type of storage, the facility owner gets a much better value from the building. SBT suggests that the owner work with its suppliers to have them deliver as needed instead of in bulk.
The plant will require water, and estimated 100,000 liters per day from an on site bore hole, a 1,500 amp electrical service, note that alternate energy sources are being investigated, wind and solar, but are not included in this proposal. Natural gas to be used by a standby electric generator and to power the steam generators and still would be good but not necessary. If the location chosen can support wind turbines they should be considered. They are not included in the cost of the project. See power load attachment.
The quality system used in the production of IV solutions is very comprehensive in that it conforms to the EU requirements and ISO 9000 requirements.
Each area of the production process has specific checks that are performed for each batch being manufactured. Manufacturer uses a specially designed bar code system, developed for the manufacturer by Valuetrak. The system is networked to the management information system for purchasing support, reporting any problems that involve a product supplied by a contracted provider plus numerous records required for validation of the products.
A purchase order is issued for a specific chemical, in the purchase order the exact compound is specified with both the word name and chemical formula. When the product is received at the plant the labeling of the container, is compared with the chemical name and formula provided on the shipping document to the purchase order. All this date is entered into the system. Also, on chemicals, a random sample(s) is taken from each lot number of the shipment and analyzed in the lab, confirming the correct chemical is in the container shipped from the supplier.
When chemicals are prepared for a production batch the information is recorded on which chemicals, they are weighed per the formulary, mixed dry, in most cases, and the final mix is sampled, verified in the QA lab before being put into the mixing tank. Depending upon the formulary for the production the chemicals are diluted with WFI water to a certain level. Again the concentration is checked in the QA lab. If the mixture conforms to the formulary the final dilution is then done in the tanks. After the final dilution is completed the final concentration in the solution is tested in the QA lab. If correct the solutions is release for production. Corrections can be made to the solutions to bring them into conformity with the formulary at any point up to the release for production.
Once the solution mixture is released for production all the data is stored in the QA file on that batch. This information has to be maintained for a minimum of 7 years so the file is encrypted preventing any changes.
The plastic raw material used in the production of the containers follows a similar process as the chemicals except the lab probably cannot chemically analyze the plastic pellets. An alternate method of verification of the pellets would be used based on weight and conductivity.
On the water side of the production testing is done on the “raw” water coming into the system for the levels of minerals found in the initial tests plus any additional hydrocarbons that may have been introduced into the supply by outside sources. This testing includes the pH level of the water. After the pre-treatment step the water is again tested to be sure the hydrocarbons have been removed and the pH balanced to the needs of the formulary to be produced.
After the R/O step, the water is again tested to be sure that the minerals have been removed or reduced to a level that they do no effect the formulary.
The steam generator is constantly monitors for temperature and pressures while the output of the still in monitored via resistance measurements to assure the water is WFI quality. Water that does not meet the WFI standard is returned to the input for additional processing.
All monitored parameters are automatically entered into the QA database along with any derived or computed information from various instruments. This document is tagged with the batch number and combined with the chemical information described above into an encrypted record.
The products are sampled per validation requirements for consistency, including formulary of the solutions.
The autoclave is monitored for temperature, pressure and time and all automatically recorded into the database.
The final production test is the bacterial over warp that is applied to the containers before being placed into boxes for shipment to users. Here a certain number of containers will be selected for retention at the facility for a period of time as a quality standard for that batch.
Again all the monitored steps are included automatically into the database and computed/measured steps by humans are entered.
The manufacturer will also provide the owner/operator with the following documents:
All manuals of equipment and equipment specifications
All GMP standard operating procedures (SOPs)
All maintenance standards and operating procedures
Production data such as drawings, lists, diagrams, and functional descriptions
All quality control and assurance manuals
All maintenance manuals and maintenance schedules.
The Good Manufacturing Practice (GMP) includes the Standard Operating Procedures (SOP) for the production equipment, and the preventative maintenance requirements for the equipment will be provided. Both are in a format that can be presented to the regulating authorities for medical devices in the U.A.E..
The validation documentation includes the documents for each production device (from the manufacturer) and quality requirements for all products used in the production of solutions, and test procedures.
All equipment is connected to the computer system so all steps are systematically monitored and reports generated. The manufacturer will be able to query the computer from its corporate and/or regional office to monitor any production problems and suggest any necessary corrective actions to maintain the quality level of the products..
When the installation is completed, Manufacturer personnel will train the owner/operator’s staff on how to operate the machines, follow procedures, create reports, conduct maintenance of the machines, and prepare validation documents as needed. It is projected that the manufacturer will provide a training staff of 1 to 4 people for 1 to 8 weeks, dependent on the knowledge of the staff members and how quickly the staff becomes comfortable with the processes. Training will be conducted exclusively in English. The services of a translator, if desired, will be paid for by the Owner. All training will be done at the project site.
Two of the employees should have good mechanical skills, as they will be trained on the Form Fill Seal (FFS) machines. They will be taught how to change Welding dies, (the welding dies control the size of the bags being produced), machine programming, how to repair and calibrate the units, they will participate in the final testing of the units at installation and review all the documentation. They also will be trained on the water treatment system. They will be taught how to change filters and membranes, repair and calibration of the machinery, its operation and documentation of their actions.
Two employees with chemistry background will be trained on the laboratory equipment, formulary of the solutions to be produced and quality assurance for the product line. These are very key positions as they will be controlling the quality of the product produced and will be required to handle all of the documentation on the product purity and consistency.
All of the employees must be involved with the equipment installation. During the installation and pilot runs, engineers from the manufacturer and the various suppliers will be on site for the final testing and validation of the plant. Additional personnel will be trained during this process.
Manufacturer will also provide training to the warehouse personnel, machine operators and all other manufacturing and quality control personnel.
The cost of the above training, except for the salaries of the owner’s workers is paid by the manufacturer.
Manufacturer will be responsible for overall project management, including, but not limited to, engineering, ordering, shipping, erection, training, validation, and certification. Manufacturer will work with the client to retrofit an existing building or provide designs for a new one. Manufacturer will keep a project team onsite at all times during the installation and certification process.
Initial project tasks include:
INFORMATION NEEDED BEFORE FINAL DESIGN
water sample
building code
electrical code
any local laws that will effect production or sales
electrical service
sewer
PERMITS Please note that are permits must be obtained and paid for by the owner
building permit
business permit
banking permit
permit to hire workers for construction
insurance
SHIPPING INFORMATION
what port
receiving agent
customs agent
required documentation
The standard of the Building and equipment therein will in principal follow Current Good Manufacturing Practices GMP for Pharmaceuticals
The detailed design of the Plant will be done by utilizing manufacturer’s experience and know-how from similar projects to determine the placement of equipment, drains, utility connections, electrical panels, computer wiring, piping, and communication systems.
Manufacturer will supervise the construction process, via a video connection and act as a Consultant for the client to make sure that the main contractor (appointed by the owner/client) follows the agreed upon specifications, design and drawings.
Manufacturer will supply all equipment and machinery (100%) necessary for a fully operational, functional and capable facility from international suppliers that comply with the EU and ISO 9000 requirements for product quality
The main equipment will include (but is not limited to):
Water preparation
Mixing & preparation
FFS packaging machine
Sterilization
In-process laboratory
QC Lab
Computer network and system
All equipment, machines, items and systems are warranted for one year from the date of the first beneficial use by owner, which is to say from first certified production.
Manufacturer will arrange for all necessary and appropriate packaging and shipping of the facility components to the designated debarkation port in the local country or region. All items will be insured during the shipping time.
Shipping costs shall be paid by the owner and manufacturer will work with the suppliers to consolidate equipment in an effort to reduce shipping costs.
All items, equipment and machines will be inspected before shipping and/or in the original manufacturer's sites.
Manufacturer will be fully responsible for the installation and commissioning of ALL equipment, machines, items and systems.
Manufacturer personnel and engineers will also select and appoint qualified local tradesmen to assist with the installation process on jobs such as plumbing, communication and machines wiring etc. All these costs are included in this proposal and will be paid directly by the manufacturer.
Master files for the most common IV solutions used in healthcare will be provided by the manufacturer.
These files will include the following:
- Composition
- Specification of the finished product
- Specification of the raw materials
- Specification of the packaging materials including labels
- Instructions
- Test methods for the finished product
- Test methods for the raw materials
- Test methods for the packaging materials including labels
- Manufacturing methods and Batch Production Record
- Stability Studies
Manufacturer will also provide the owner/operator with the following documents:
All manuals of equipment and equipment specifications
All GMP standard operating procedures (SOPs)
All maintenance standards and operating procedures
Production data such as drawings, lists, diagrams, and functional descriptions
All quality control and assurance manuals
All maintenance manuals and maintenance schedules.
The Good Manufacturing Practice (GMP) includes the Standard Operating Procedures (SOP) for the production equipment, and the preventative maintenance requirements for the equipment will be provided. Both are in a format that can be presented to the regulating authorities for medical.
The validation documentation includes the documents for each production device (from the manufacturer) and quality requirements for all products used in the production of solutions, and test procedures.
All equipment is connected to the computer system so all steps are systematically monitored and reports generated. Manufacturer will be able to query the computer from its corporate and/or regional office to monitor any production problems and suggest any necessary corrective actions to maintain the quality level of the products.
Manufacturer will provide the owner with the required qualifications of all necessary employees and personnel needed for the facility to operate at full capacity.
Manufacturer will assist the owner in the selection, interviewing, and hiring of all initial employees.
Barring civil disorders or acts of God, the Project will be complete in 12 – 16 months.
1- Signing MOU/contract between the manufacturer & the Investor/owner and a payment or opening of an LC
2- Opening the Final LC to the manufacturer, with terms and targets to start the project along with a draw down schedule for the funds..
The owner is responsible for all permits and government fees that may be required
The Production machinery, to produce up to18,000,000 units (500mL) per year (based upon a 312 day production schedule), shall include the following components:
1 Plumat Model FFS-3000 form/fill/seal machines with Model OW 834 overwrap machine
Clean in place steam systems to sterilize the machines between production batches
Visual inspections systems, which allows the operators to visually check every unit as it comes out of the machine.
Automatic leak detector systems, which tests for leaks in the containers. If leaks are found, it rejects those containers and notifies the operators of a potential problem.
1 10˚ C chilled water system, at a flow rate of 16 liters per minute, including
heat exchanger
1 Air compressor, oil-less sized to meet the needs of 4 machines
1 Class 10,000 clean room, hard wall with air locks and HEPA filters
Production support equipment provided includes:
1 Water treatment system that process up to 60,000 liters of processed water per day including 40,000 liters of WFI to be used in the production of IV solutions. The remaining water is to be used for, sterile steam, cleaning and general use within facility.
2 30,000 liter stainless steel jacketed tanks to hold the solutions prior to packaging with stirrer to keep the solutions in suspension
The Sterilizer/autoclave section contains the following:
1 Up to a 3 cubic meter dual door autoclave with computer controls,
3 Product racks to load the solutions containers on before movement into the autoclave
3 Carts to move the product racks into and out of the autoclave
Quality Control Laboratory
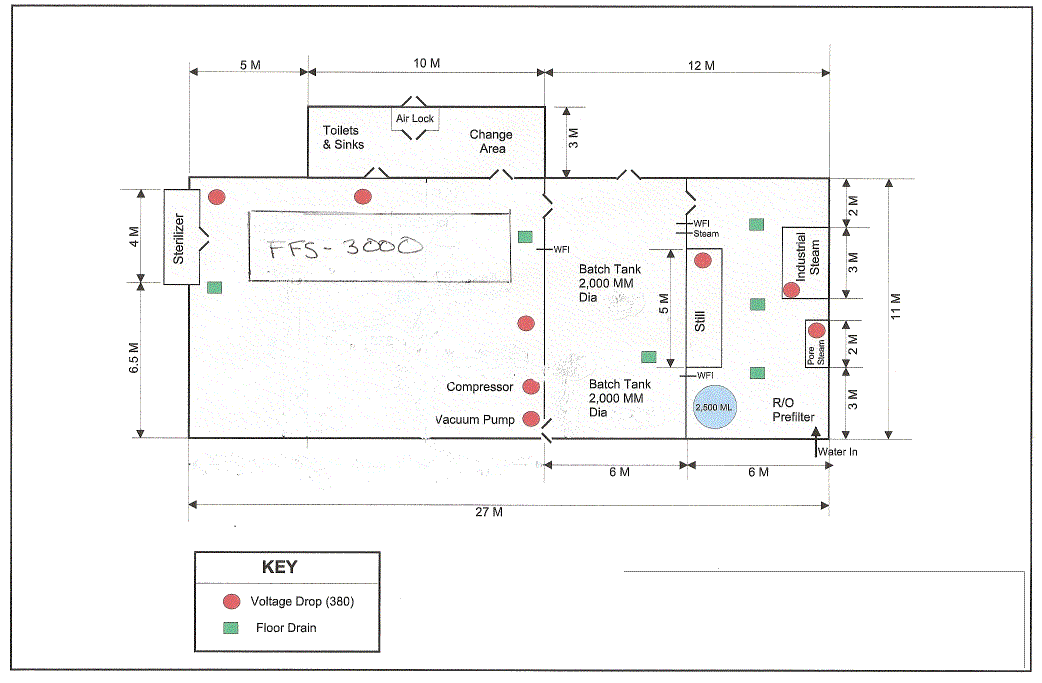
As part of the building, a separate laboratory area must be created. This area should have floor space of 30 square meters and be on an outside wall so the fume hood can be easily vented. The space should be under positive pressure at all times from the ventilation system.
1 Liquid chromatograph, Agilent model A2000 or equivalent
A/N Additional columns as determined by the formulary selected and water quality testing
1 pH meter, Orion or equivalent
1 Bench top incubator for bacterial testing
1 Microscope with light source
1 Vented fume hood
1 Bench top centrifuge, 8 station variable speed
1 Safety cabinet
A/N Test tubes, culture trays, glassware
1 Laboratory grade electronic scale, accurate to 0.05 grams
1 Mixer for solutions
1 Hotplate/water bath
1 Electronic thermometer
A/N Basic supplies for both wet and dry testing
Main Quality Control Laboratory Equipment
As an essential part of the quality assurance program, a testing laboratory is included in the turnkey project. This area is completely separate from any of the manufacturing space, and should be at least 30 square meters and be on an outside wall so the fume hood can be easily vented. The space should be under positive pressure at all times from the ventilation system.
Building specifications for IV solution plant with additional plastic production capabilities.
Basic building
Outside dimensions 36 by 70 meters with 5 meter high wall
First floor 2,500 square meters of space
Construction type Multiple Frame Steel
Roof and side walls 26 gage, 80,000 PSI yield strength, with zinc and aluminum coating in the choice of 8 colors.
Insulation 10CM vinyl backed fiberglass, in roof and walls
Ceilings first floor open to underside of roof except in clean room area and office/lab space, ceiling to be determined
Air conditioning warehouse area by fans
Clean room, office and lab central air
Windows as desired up to 60
Shipping Door 2 electrically operated
Entrance doors as required by building codes
Rest Rooms as required by building codes, estimated 20 people per shift in the building
Lighting Production floor, metal halide, other areas to be determined
Electrical outlets as required
Electrical panels Main Power 1500 amp, 440 volt three phase 50Hz
Sub-panels Blow fill seal machine
Water treatment
Injection molding machines
Sterilizers
Laboratory
Office space
Electrical distribution in metal conduits to machines, plastic/rubber jacket in walls of office space and laboratory.
Electrical ground as required by local code, suggest saline ground rod
Heating system scavenged and waste heat from still and sterilizer distributed via metal air ducts
Floor Drains under blow fill seal unit, tanks and water treatment areas, outlet to be determined, (dry well, sewer or septic system)
Waste water need to be handled by either an on site septic system or the municipal sewer system.
Fresh water source to be determined, well or municipal
Foundation as required by local building code
Footings 1 square meter by 1.5 meters deep at each corner of the building, plus 0.5 square meter by 1.5 meters deep at each building truss, (every 10 meters)
First floor 15 CM thick re-enforced concrete floor with sealer coating, flatness of the floor to be determined.
Internal build out The price quoted covers the building of office and laboratory space in the building. The wallboard, studs and wall sealing material will be shipped with the building. Local craftsmen will be employed to do the work but under the direction and with the tools supplied.
Phone and data cables as required, supplied with building
General notes 1 The shipping doors should open onto a platform that is tailgate height for trucks that will deliver raw material and ship out finished products. This will effect the grading of the land or the foundation of the building.
2 The shape of the plot and any slope may effect where the
shipping doors can be located in the building
3 It is expected that trailers of up to 17 meters in length
may either deliver or pick up material from the plant so space for trucks of that size to maneuver has to be planned.
4 The car parking requirements for the plant have to be determined.
| Power Load IV Production Plant | FFS 3000 | ||||||||
| Device | Voltage | Phase | Peak Load | Running Load | Watts | AC Factor | Draw Time | kWh | |
| amps | amps | ||||||||
| FFS 3000 System | 380 | 3 | 65 | 50 | 2200 | 2530 | 16 | hours per day | 304,00 |
| Air Compressor/Vacuum Pump | 380 | 3 | 17 | 12 | 4560 | 5244 | 12 | hours per day | 54,72 |
| Hear Exchanger | 380 | 3 | 32 | 25 | 1450 | 1667,5 | 16 | hours per day | 152,00 |
| Mixer | 220 | 1 | 4 | 3 | 660 | 759 | 4 | hours per day | 2,64 |
| Scale | 220 | 1 | 1 | 1 | 220 | 253 | 2 | hours per day | 0,44 |
| WFI Pumps | 220 | 1 | 14 | 10 | 2200 | 2530 | 8 | hours per day | 17,60 |
| Autoclave | 380 | 3 | 35 | 20 | 7600 | 8740 | 24 | hours per day | 182,40 |
| Computer system with printers | 220 | 1 | 4 | 2 | 440 | 506 | 24 | hours per day | 10,56 |
| Laboratory equipment | 220 | 1 | 16 | 10 | 2200 | 2530 | 8 | hours per day | 17,60 |
| Clean room | 380 | 3 | 50 | 30 | 1500 | 1725 | 24 | hours per day | 273,60 |
| Remaining space | 220 | 1 | 20 | 15 | 3300 | 3795 | 24 | hours per day | 79,20 |
| Still/Boiler | 380 | 3 | 25 | 18 | 335 | 385,25 | 16 | hours per day | 109,44 |
| totals | 1204,20 | ||||||||
| These numbers assume that the still and stream boilers are not run with electricity, if they are we would need and addition 400 amps at 380 | |||||||||
| phase for 16 hours per day dropping to 100 amps for 8 hours when the still is not in use. | |||||||||
| kWh= Volts x amps x Draw Time / 1000 | |||||||||
Specification and photos are not contractual and are subject to verification upon inspection
TAKE NOTICE!
PLEASE BE ADVISED THAT INFORMATION INCLUDED IS CONFIDENTIAL IN NATURE AND IS BASED ON PRE-EXISTING BUSINESS RELATIONSHIP WITH THE LEGAL OWNER OF PROPERTY DESCRIBED HEREIN (IF APPLICABLE). AS SUCH, UPON RECEIPT OF SAID INFORMATION THE RECEIVER ACKNOWLEDGES THAT ANY UNAUTHORIZED CONTACT WITH SAID LEGAL SELLER WILL BE CHARACTERIZED AS A BREACH OF CONFIDENTIALITY AND SAID AGREEMENT MAY BE ENFORCED UNDER EXISTING LAW OR IN EQUITY.
This paper was
prepared by General Equipment Inc.
The paper represents an offer of a partner of General Equipment Inc.
All rights are reserved by and for General Equipment Inc.
All
content and ideas of this paper are the property of General Equipment Inc.